
Regulations and Considerations for Marketing Heat Patches
Release time:2023-10-10 Click:129
Heat patch manufacturers and marketers should adhere to relevant regulations including:
Medical device marketing guidelines on permissible claims and required warnings and disclaimers.
Regional regulations governing approval of OTC topical pain medications which may classify patches as drugs.
Testing and reporting requirements set by oversight bodies for safety and performance verification.
Responsible advertisement guidelines regarding clear communication of risks and avoidance of inappropriate promotions.
Substantiation for claims of specific benefits like improved circulation through clinical studies.
Protocols for reporting of adverse effects observed in patch users after sale.
GMP standards and quality systems for consistent, high quality patch production.
A strong understanding of regulations helps heat patch brands bring compliant, low-risk products successfully to global marketplaces.
Hot products

CBD Pain Relief Patch
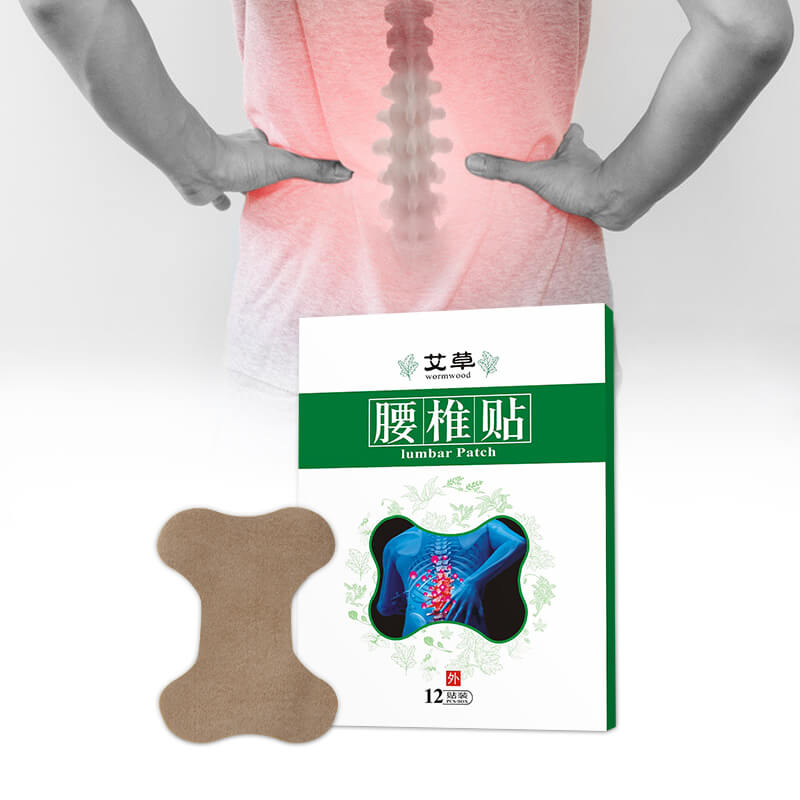
Lumbar Plaster Pain Relief Patch

Lavender Detox Foot Patch
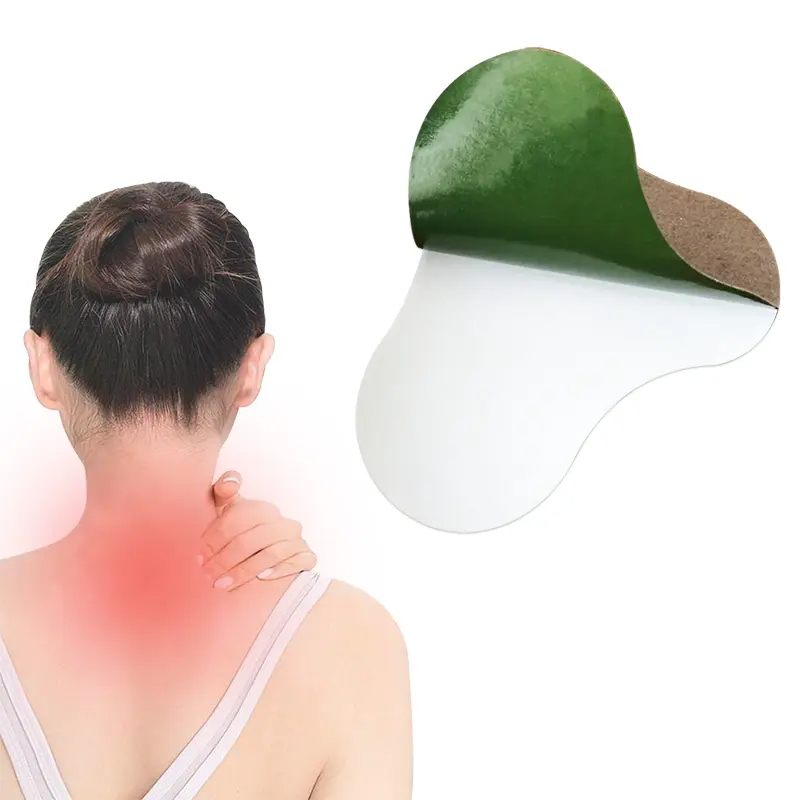
Cervical Vertebra Pain Relief Patch
Tel:0086-18939260319
E-mail:kongdycn201810@gmail.com
